AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Pdfinfo water chemistry3/10/2023  Determining a reliable average depends on the standard deviation (SD) of the measurements, with more measurements required if the SD is larger. 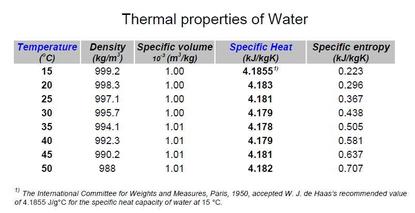
When it is crucial to accurately determine true average As concentrations – for example, at one of the many wells that fluctuate above and below the regulatory As limit of 10μg/L – it is worthwhile collecting samples frequently during pumping to more accurately determine the average As concentration. The As concentration variability is likely due to As adsorption reactions between Fe oxides in the well borehole and in the aquifer near the well borehole during periods when the pump is not in operation. In these wells, the As concentration decreased to below 10μg/L again when pumping was briefly restarted after being stopped for 4h. In these wells, the As concentration was less than 10μg/L shortly after pumping started, but the As concentration increased over time to a level exceeding 10μg/L. In some cases, significant As concentration variability was noted over a short period of time. Results of intensive sampling conducted over the course of approximately 1 year at 3 public water system wells is consistent with the analysis of historic As measurements. The average As concentration is commonly within one standard deviation of the MCL. Arsenic concentrations fluctuate above and below the USA’s As drinking water Maximum Contaminant Level (MCL) of 10μg/L. There is significant random and systematic variability in As concentrations in numerous public water system wells in Minnesota. While surface solid analysis did not provide conclusive evidence confirming the nature of orthophosphate?s control mechanisms, jar tests and equilibrium solubility models were demonstrated to provide useful quantitative predictions of how orthophosphate reduces copper concentrations in various waters. This field research generally supports results previously reported in the literature: in high alkalinity, neutral pH water, a dosage of 3 ? 4 mg/L orthophosphate can reduce copper levels in a drinking water system from over 2 mg/L to below the 1.3 mg/L USEPA action level. This study analyzed field data obtained over the course of a year from a high alkalinity water system into which orthophosphate was added to control copper concentrations. The research questions were answered through a comprehensive literature review and experimental methodology integrating laboratory jar tests, water sampling and analysis from a field investigation, qualitative solids analysis, and equilibrium model application. 
Specifically, this thesis sought to answer three research questions regarding the impact of orthophosphate treatment, the nature of the mechanism by which orthophosphate controls copper corrosion, and the value of equilibrium modeling in predicting orthophosphate?s effects. The purpose of this research was to investigate orthophosphate as a corrosion inhibitor for copper pipe in a high-alkalinity drinking water system. Model results show that PAB is effective for the remediation of a PCE-contaminated aquifer, since the concentration of PCE flowing out of the barrier is everywhere always lower than the concentration limit provided for in the Italian regulations on groundwater quality. 
A case study in which the procedure developed has been applied to a tetrachloroethylene (PCE)-contaminated aquifer near a solid waste landfill, in the district of Napoli (Italy), is also presented and the main dimensions of the barrier (length and width) have been evaluated. The complete characterization of the chemical-physical interactions between adsorbing solids and the contaminated water, required by the computer code, has been obtained by experimental measurements. A computer code, including different routines that describe the groundwater contaminant transport and the pollutant capture by adsorption in unsteady conditions over the barrier solid surface, has been developed. A procedure to optimize the design of a Permeable Adsorptive Barrier (PAB) for the remediation of a contaminated aquifer is presented in this paper. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
